Introducing some new friends
in the atrium
Seems like yesterday since I came to Karlsruhe, but here we are, 2020 is almost done and I am about to complete the third month of my PhD.
A lot of things have changed during these months. For example, I ride my bike to go everywhere, no matter if it’s just around the corner or in a different town. However, some other things have sadly not changed so much: we are still under lockdown and our “social life” is restricted to the time we spend together at KIT. But of course, we should look at the bright side and think that we are safe and sound!

And one of the “brightest things” in this time of the year is, of course, Christmas lights. I have always been a huge fan of them (although I really agree there are thousands of things in which we should invest the money instead) and being in a new city always means a Christmas walk with my camera. I really enjoy the streets of Karlsruhe covered with thousands of tiny shiny lights, the environment of Christmas markets, passers-by with their Glühwein (basically hot wine with some cinnamon flavor) and kids playing around Christmas trees. For a moment you can really forget about all this “pandemic situation” and that is something that only Christmas spirit can do.

Speaking of forgetting about this situation, if you remember for my first post, we were in the middle of our first Summer School in Data Mining and Statistics. Although it was quite intense and a lot of work, I am really forward to having the next one (hopefully in person!) and meet all these guys “face-to-beer”.
However, it meant I didn’t have much time left to discover the following path of my research project, so after the Summer School had ended, I really started sailing into the world of AF. To give all of you a general overview of my project, I am going to be inspired by some voice notes I am sending to my closest friends to keep them aware of the “weird things” I do as part of my PhD:
My project is part of the Working Package 1, which is related to technologies for characterizing the atrial state. In the heart, regions of scar and different levels of fibrotic tissue have been identified as potential driving regions of arrhythmias. Nowadays, these regions can be characterized by magnetic resonance or echocardiographic imaging among other tools, but it might be also possible to use impedance measurements to quantify atrial fibrosis. This system could also be implemented on intracavitary recordings and help detecting atrial substrate that promotes onset and maintenance of atrial fibrillation and, that way, improving the success of ablation strategies.
But… wait, wait, wait. Ablation? Substrate? Atrial fibrillation? What are you even talking about?!
There are some companies working on catheter developments and investigation. That is a fact. Well, these catheters are used for performing ablations in Atrial Fibrillation (AF).
But what does that actually mean?
As many of my colleagues have previously introduced in some of their posts, “AF is the most common cardiac arrhythmia, which generates a general disorganized electrical and mechanical activity on the atria with a mechanism of multiple re-entrant wavelets” (https://www.ncbi.nlm.nih.gov/books/NBK2219/). To alleviate all this chaotic heart activity, ablations are performed. Ablation consists of burning an area of cardiac tissue that is causing problems in transmitting heartbeats and therefore helps to maintain this chaotic activity. This is done by burning the area of the pulmonary veins (PVI: pulmonary vein isolation) and the order of the heart is more or less restored.
But how do we get to the heart?
Well, thanks to the blood vessels that run through our whole body we can access the heart through these “pipes”. Using the Seldinger technique (http://cardiaccathpro.com/VascularAccess.html) it is possible to get into the left atrium of the heart and place catheters inside in order to perform the procedure.
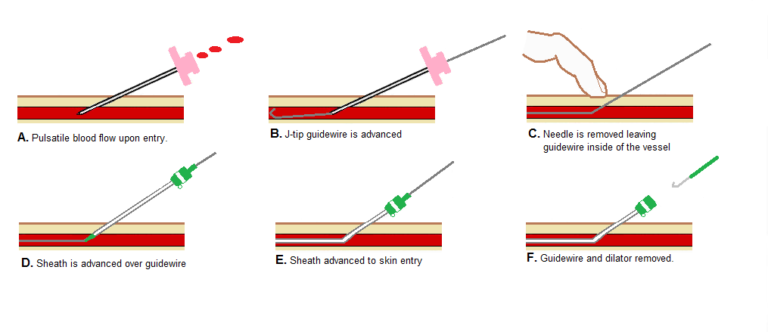
As we have already said, we need a catheter that not only is capable of carrying out the ablation but also to measure impedance. But first, we would need to know “where are you exactly inside the heart” because otherwise, it is impossible to guess which parts should be ablated. Thus, another catheter is mapping the atria from inside being in contact with the walls of it. For instance, it is like people are inside a dark room and they want to know the room’s shape, so they start touching the wall until everything is “mapped”. So it is with the atrium! That way, we know the limits of the atrial wall and it is possible to characterize the tissue.
So far so good, keep going!
On the other hand, the impedance catheter needs a sheath to help its stability. However, if the sheath overlaps with one or more of the catheter electrodes, distortions can occur and not be noticed. So, I am currently working on understanding one of the most commonly observed artifacts which occurs when the catheter enters too far into the sheath. Overall, we are trying to understand the nature of all kinds of artifacts in the clinical impedance recordings with the help of computational modelling and experimental data.

Und das ist alles! (And this is all!) I really hope you learnt a little bit more about how AF is treated and how these methods can be improved for better measurements. In my next post (probably written from a different country!) I will explain how impedance works in the heart. Have a nice month, a wonderful entry in the new year and stay tuned!
